May 15, 2023
What has changed
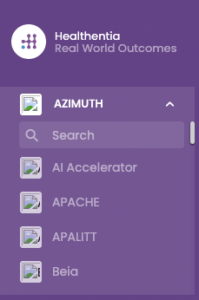
We have improved the study selection menu on the left ordering the studies alphabetically and including a search bar for easier search.
How it affects you
Easier navigation from study to study.
What has changed
We have improved the study selection menu on the left ordering the studies alphabetically and including a search bar for easier search.

How it affects you
Easier navigation from study to study.